Radiation Therapy Treatment
What to expect during radiation treatments
Your specific radiation therapy treatment will be determined following a simulation appointment that includes a non-diagnostic CT scan of the area to be treated with radiation.
Your physician will consider the type and size of the cancer, location in the body, proximity of the tumor to normal tissues that are sensitive to radiation, your general health and medical history, and any other cancer treatment you may be receiving, such as chemotherapy.
Contact Us

Florida Cancer Specialists & Research Institute (FCS) is accredited by the American College of Radiology (ACR) for radiation oncology. FCS earned a three-year accreditation for Radiation Oncology Services. The prestigious ACR Gold-seal designation is awarded to all FCS sites of service.
Frequently Asked Questions
FCS offers several types of radiotherapy treatments including:
- AlignRT/Surface Guided Radiotherapy (SGRT)
- Brachytherapy
- Breast Brachytherapy (SAVI)
- Image Guided Radiotherapy (IGRT)
- Intensity Modulated Radiation Therapy (IMRT)
- On-Board Imager
- Stereotactic Radiosurgery (SRS)
Click here for more detailed information on the types of radiotherapy available at FCS.
What is radiation therapy?
Radiation therapy, also known as radiotherapy, is a treatment that uses high-energy radiation to kill cancer cells or shrink tumors. It works by damaging the DNA inside the cancer cells, preventing them from multiplying and growing. Radiation therapy can be delivered externally using machines outside the body or internally by placing radioactive materials directly into or near the tumor. Radiation therapy is often used in combination with other cancer treatments like surgery and chemotherapy.
Back to Top
What are the benefits of radiation?
What are the benefits of radiation? The goals of radiation therapy are varied and depend on the patient and their cancer. Your physician may recommend radiation therapy for any of the following reasons:
- To cure or shrink your early‑stage cancer
- To prevent cancer from coming back in other parts of the body
- To treat symptoms caused by advanced cancer
- To treat cancer that has returned
Who performs radiation treatments?
Delivery of radiation therapy is provided by a team if specialists that includes:
- Radiation Oncology Physician, who evaluates and determines the appropriate therapy or combination of therapies based on the area receiving treatment.
- Therapeutic Medical Physicist and Dosimetrists – together with your radiation oncologist, they perform detailed treatment calculations
- Radiation Therapists are specifically trained to acquire images and deliver the radiation treatments.
- Nursing Professionals work with the team to assess your needs, provide education and help manage any reactions or side effects that may occur during treatment.
What are the side effects of radiation treatment?
The side effects of radiation therapy vary and depend upon the treatment dose and the part of your body being treated. Possible side effects include fatigue, skin irritation, hair loss and loss of appetite. Your FCS care team will help guide you through your treatment and offer tips for managing side effects.
Back to Top
How do radiation therapy treatments work?
Radiation therapy involves the use of high energy x-ray beams. Radiation shrinks tumors by destroying cancer cells. FCS uses the most advanced three-dimensional planning, which allows precision and versatility for all therapeutic application. Virtually all cancers respond to this treatment. Pain and other cancer related symptoms can also be reduced.
Back to Top
How long will each radiation treatment last?
Most courses of radiation therapy require 25 to 35 treatments over a five-to-seven-week period. The length and intensity of therapy can vary, depending on the type of cancer and your response to treatment. In addition to the radiation treatment sessions, our patients typically have one visit with a radiation oncologist each week while they are receiving treatments.
Back to Top
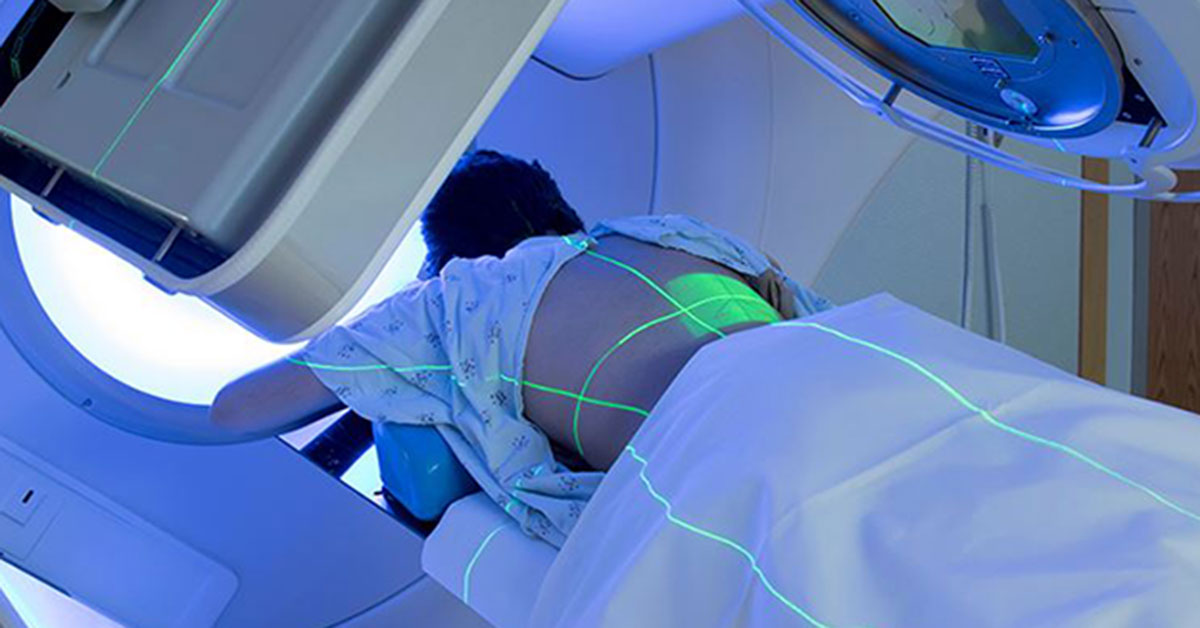
What will I feel during and after the procedure?
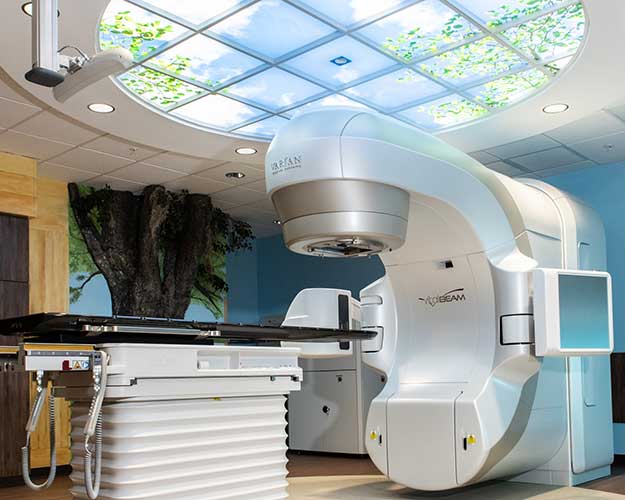
Throughout the imaging and radiation therapy session you may see and hear the equipment rotate around you, however, the sessions are painless. The linear accelerators from Varian Medical Systems combine radiotherapy treatment technology with sophisticated imaging capabilities for precise forms of image guided radiotherapy. This technology allows the physician and therapist to visualize and verify that treatment is applied to exact area.
Back to Top
What is a linear accelerator?
A linear accelerator (LINAC) is a computerized machine used for external beam radiation treatments. The LINAC is used to treat all areas of the body by delivering high-energy x-rays to the area of the patient’s tumor. By design, they destroy cancer cells while sparing surrounding normal tissue. The linear accelerator uses electricity to produce high energy x-rays. The x-rays are planned to exit the machine in the shape of each patient’s tumor and are directed to the patient’s tumor. The beam exits the machine through the gantry which can rotate around the patient.
Back to Top
What is the difference between radiation therapy and chemotherapy?
The main difference between radiation therapy and chemotherapy lies in how they work and how they are administered:
Radiation therapy uses high energy radiation to target and destroy cancer cells or shrink tumors. Chemotherapy involves the use of drugs to kill cancer cells or inhibit their growth by interfering with their ability to divide and multiply.
Radiation therapy targets specific areas of the body where cancer is located or where there is a risk of recurrence. Chemotherapy is administered intravenously or orally and circulates throughout the body, targeting cancer cells wherever they may be, including those that may have metastasized to other parts of the body.
Back to Top